Use of this equipment adjacent to or stacked with other equipment should be avoided because added pressure could result in accidental damage. If stacking of equipment becomes necessary, The Fischer and any other equipment should be observed to verify that they are operating normally.
Place The Fischer and water bath trays on a firm, level surface.
Please always cover the treatment electrodes with the foam inserts or sponge pads provided; otherwise, you might risk burns (electrical burn injuries, current marks). Avoid direct contact with the electrodes.
Do not use the wearable parts (foam inserts and sponge pads) for more than 18-months and replace them on time.
Make sure that The Fischer is stored at room temperature before you power it up. Depending on storage conditions, the temperature adaptation may take up to 2 hours.
Prior to starting a treatment session, please remove any metallic jewelry (rings, watches, bangles etc.), as electric current concentrations in these regions might result in minor injuries (local electrical burns, current marks).
Any small skin lesions or breaks must be covered with petroleum jelly (Vaseline) to insulate them from the current flow.
Depending on the current strength selected, users may experience tingling or stinging sensations (discomfort) during the treatment.
If the current is set too high, you may experience painful sensations all over the treated extremities.
You may remove your hands or feet safely from the treatment trays any time. In very rare cases, unpleasant electric shocks may occur; these are, however, absolutely harmless.
During the first treatments, you may experience increased sweat production; these symptoms will spontaneously abate after the next few treatment sessions.
The treated skin can show signs of dehydration like flaking or small lesions. Should this be the case, please apply a rehydrating cream after treatment.
Immediately following a treatment session, occasional redness may be observed. This is temporary and a sign that the treatment is working (stimulating capillaries). In rare cases, blistering may occur. Reducing the treatment intensity can minimize the appearance of these symptoms. Before administering a new treatment session, the skin has to heal completely. In case of an unexpectedly strong skin reaction, a medical professional must be consulted before further treatments are administered.
You may not be treated by two devices simultaneously. The device may not be modified.
Keep out of reach of children unless otherwise supervised. Do not leave the device unattended. Risk of strangulation and injuries.
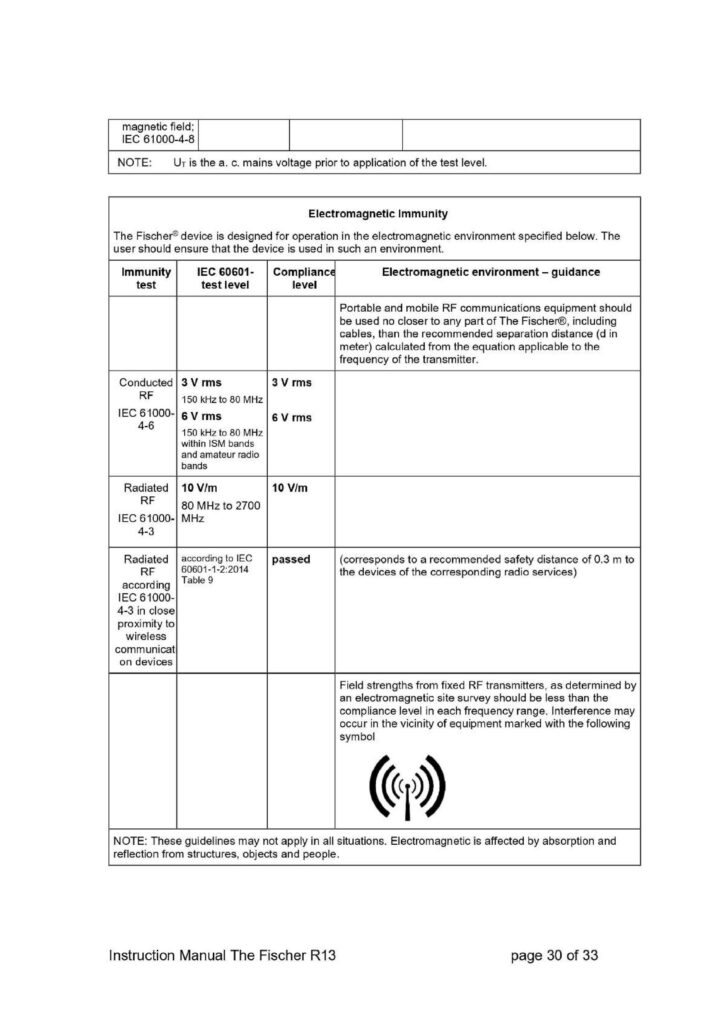
Portable RF communications equipment (including peripherals such as antenna cables and external antennas) should be used no closer than 30 cm (12 inches) to any part of the device, including cables specified by the manufacturer. Otherwise, degradation of the performance of The Fischer could result.
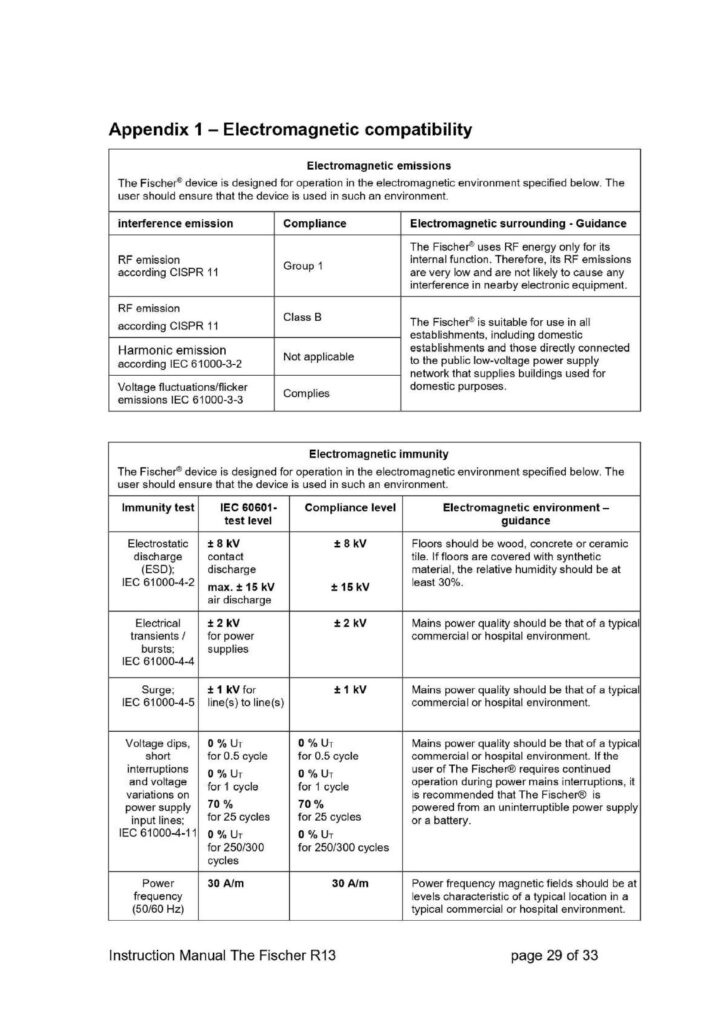
Follow the recommended separation distances listed in appendix 1.
Disconnect the power supply from the socket if a thunderstorm approaches or if you do not plan to use The Fischer for an extended period of time.
Position the device so that you can unplug the power supply unit from the socket anytime.
The Fischer is designed for indoor use only. Do not expose it to rain or humidity.
If you wish to clean The Fischer, pull all plugs from the socket and switch it off. Use a soft, damp cloth and a mild cleaning solution to clean the device.
Make sure you do not pull the cable too hard and do not expose the cable to heat or chemicals. If a cable is damaged, unplug it from device or electrode, respectively, and have it checked by RA Fischer Co.
Do not open the device. There are no control elements inside. The treatment system may only be serviced by RA Fischer Co.
Warning: Potential systemic adverse effects may result from use of this device. Drugs or solutions delivered with this device have the potential to reach the blood stream and cause systemic effects. Carefully read all labelling of the drug or solution used with this device to understand all potential adverse effects and to ensure appropriate dosing information. If systemic manifestations occur, refer to the drug or solution labelling for appropriate action.
Warning: Do not use when a thunderstorm is in the vicinity.
Warning: The long-term effects of chronic electrical stimulation have not been established.
According to EN60601-1-2 The Fischer device does not have any essential performance characteristics that are not related to the basic safety.
Requirements for:
Operation
Transport and Storage
Relative Humidity
30 % to 70 %
< 90 %, non-condensing
Atmospheric Pressure
700 hPa to 1060 hPa
700 hPa to 1060 hPa
The device may only be used with the power supply unit that corresponds with the power supply specified on the rating plate. To ensure safe operation The Fischer, all its cables, plugs, control elements and housing components must be in faultless condition. Prior to each use, check the device for possible damage; a defective unit should not be used for treatment. Pay attention to the references on the unit and in this user manual. Switch the unit off after each treatment session.
The Fischer is a Tap Water Iontophoresis (TWI) device intended to treat hyperhidrosis (pathological sweating) affecting hands, feet, underarms, or all three. Any other use or usage beyond this scope is considered un-intended use and may have dangerous consequences.
The Fischer is intended to be used in closed rooms. Allow the device to reach room temperature before conducting a treatment. The Fischer may only be used in a dry environment. The required minimum distance to shortwave microwave devices depends on their frequency and transmission power and is defined in the Electromagnetic Compatibility tables. For additional information, please refer to item 9 “Initial Operation” in the user manual.
When starting treatment, sessions should be performed four times per week and not more than once daily for approx. 15-minutes each. After about 10 treatments, sweat secretion decreases to normal levels. Due to the reversible character of the therapy, a long-term treatment (maintenance schedule) is indicated with sessions lasting approx. 15-minutes, to be conducted once or twice per week, depending on the severity of the clinical symptoms.
Any use beyond this scope is considered “not according to intended use.” The manufacturer cannot accept any responsibility for personal injury or material damage that is or might be caused by usage beyond the scope of “intended use.”
Under no circumstances should Iontophoresis be performed if any of the following applies:
The following side effects may occur:
Should you encounter any errors prior to, during, or after a treatment session, please work through this troubleshooting checklist before contacting RA Fischer:
Rule out a device defect:
Verify that The Fischer is working by testing it without any body resistance. Prepare a water bath tray for the hand treatment as described above. Now, instead of a hand, place the second silicone-graphite electrode on the foam insert (moistened with water) and set the polarity switch to automatic. Now check the main control unit: is the timer counting down? The energy symbol flashing?
If YES, than the treatment is working and The Fischer is working properly. So why is the treatment not working when you try it? Your body resistance may be too high, or you may life in a “soft water” area, meaning your tap water lacks the mineral concentration that makes iontophoresis possible. Here’s what you can do:
In case of a device failure, or if the actions mentioned above are unsuccessful, please contact RA Fischer Co. by phone. (800) 525-3467
4 Year Warranty: The Fischer has been manufactured and tested with the greatest care. Should a malfunction occur, our manufacturer’s warranty covers all defects caused by material deficiency or manufacturing error. This manufacturer’s warranty extends over 4 years.
Any intervention on The Fischer by buyer or third parties renders the warranty null and void. Any defects which are, or might have been caused, by improper handling or disregard for the “intended use“ listed in this user manual result in the immediate loss of warranty claims against the manufacturer.
The foam inserts and sponge pads are wearable parts and thus excluded from any warranty claims. Should you, however, detect any material or manufacturing defects prior to your first use, please contact RA Fischer Co.
Repairs: Contact RA Fischer Co to begin a warranty repair.
To avoid transport damage when sending in for repair, please store the accessories for The Fischer in the water bath trays provided. If possible, ship in the original packaging. Please make sure that the device is protected against shocks and that packaging is suitable for the method of shipment selected.
In case of a malfunction, we can only take responsibility for safety, performance, and reliability of The Fischer if it is repaired by us or by persons contracted by us. Any manipulation on or repair of the device by persons not authorized by us renders our warranty and liability null and void.
Please don’t forget to clean and dry the device and accessories prior to shipping!
Please include all accessories (power supply cord, silicone-graphite electrodes, electrode cables, etc) along with The Fischer control unit.
Dimensions Control Unit
[D x W x H]
5.9” x 4.7” x 2.6”
Weight
0.66 lb
Input
Supply Voltage
Current Input
Performance Input
24 V DC
max. 130 mA
max. 3,1 W
Output Direct Current
Treatment Voltage
Treatment Current
max. 58 V=
max. 30 mA
Output Pulsed Current
Treatment Voltage
Treatment Current
Pulse Rate
max. 58 V.
max. 30 mA
10 kHz Rectangular
Protected against solid foreign objects * 12,5 mm and vertically falling water drops (power supply excluded)
IP21
Input
Input Voltage
Max. Current Input
100-240 V- / 50-60 Hz
200 mA
Output
Rated Output Voltage
Current Output: max.
Max. Output Rating
24 V=
300mA
8 VA
Requirements for:
Operation
Transport and Storage
Relative Humidity
30 % to 70 %
< 90 %, non-condensing
Atmospheric Pressure
700 hPa to 1060 hPa
700 hPa to 1060 hPa







